Global Life Sciences Update
EU Publishes Guidance on Clinical Investigations to Help Sponsors’ Compliance with MDR
The European Commission’s Medical Device Coordination Group (MDCG) recently published a new guidance (MDCG 2021-6) on clinical investigations under the MDR. The guidance is published in the form of a Q&A and may be updated with further questions and answers.
The 19-page document covers 28 questions of relevance to sponsors of clinical investigations conducted under the MDR.
The guidance covers general questions and answers, e.g., what is a clinical investigation or what is a pilot clinical investigation. For the first time, guidance is also provided into specific requirements relating to clinical investigations, such as:
- What are the general differences and improvements related to clinical investigations under the MDR as compared to the Medical Devices Directives?
- What are the safety reporting requirements for clinical investigations?
- What is the difference between the performance, clinical performance, and clinical benefit?
Regulatory pathways for clinical investigations
The guidance emphasizes that the sponsor is responsible for determining the correct regulatory pathway for its clinical investigations. It contains a helpful visual overview of the different pathways that are outlined in the MDR:
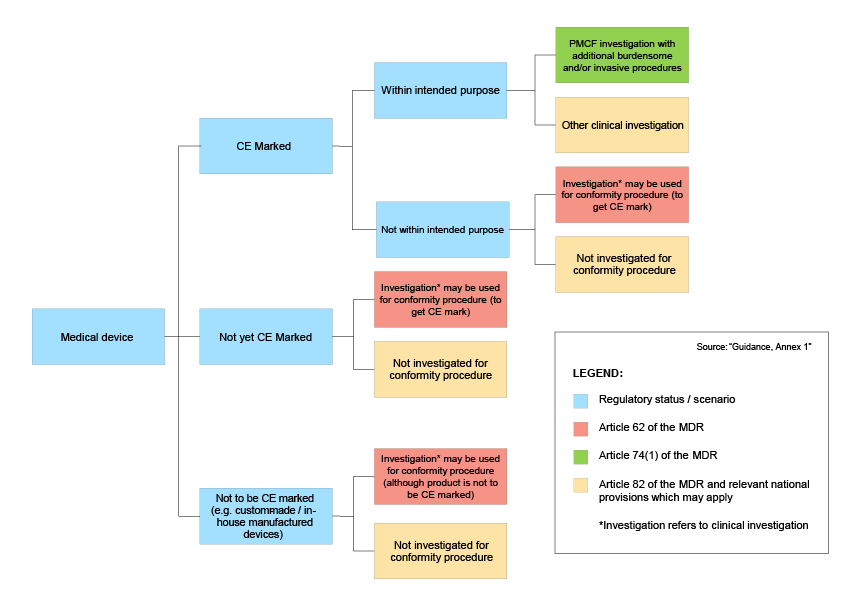
Modifications to clinical investigations
The MDR provides that if a sponsor wants to modify a clinical investigation and such modification is likely to have a substantial impact on either the safety, health, or rights of the subjects or on the robustness or reliability of the clinical data generated by the investigation, the sponsor must notify the Member State(s) concerned. The guidance helps sponsors determine when modifications are substantial, and its Annex II contains a non-exhaustive list of examples.
Annex II provides specific examples of when the following modifications are “likely” to be considered substantial:
- amendments related to the protocol or subject information,
- amendments related to the benefit/risk of the clinical investigation,
- amendments related to the use of the investigational device,
- amendments related to other information, or
- amendments relating to the manufacturing process
Burdensome or invasive procedures
Furthermore, the document provides helpful guidance on the meaning of “additional procedures which are burdensome or invasive” within the meaning of Article 74(1).
According to this Article, where a clinical investigation is conducted with an already CE-marked device, within the scope of its intended purpose, and where the investigation would subject patients to invasive or burdensome procedures, the sponsor must notify the Member State(s).
In these cases, the guidance provides the following:
- The determination of whether an additional procedure is burdensome is generally determined from the perspective of the person bearing the burden. It may include, inter alia, procedures that may cause pain, discomfort, fear, potential risks or complications/side effects, disturbances of lives and personal activities, or otherwise unpleasant experiences.
- Additional procedures that are invasive include, for example, penetration inside the body through the surface of the body, including through mucous membranes of bodily orifices, or penetration of a body cavity via a bodily orifice.
Transitional period
The guidance sets out that because the clinical investigation module in Eudamed will not be ready by May 26, 2021, sponsors should contact the national competent authorities for additional information, for example on how to submit an application for a clinical investigation under the MDR.
The guidance also clarifies the rules applicable to ongoing clinical investigations conducted in accordance with the Medical Devices Directive.
The guidance provides that these clinical investigations may continue to be conducted after May 26, 2021. However, when the MDR is fully applicable, serious adverse events and device deficiencies occurring during the clinical investigation must be notified to the Member States in accordance with Article 80 of the MDR.
The current version of the MDCG 2021-6 guidance document may be supplemented in due course with further questions and answers.
***
Manufacturers of medical devices are advised to review the guidance and familiarize themselves with the new obligations. In particular, the clarifications regarding the transitional rules and the applicable reporting obligations until the clinical investigation module in Eudamed is ready, should help sponsors of clinical investigations navigate the coming months of transition.
律师广告—Sidley Austin LLP 是一家全球性律师事务所。我们的地址及联系方式可在 www.sidley.com/en/locations/offices 查阅。
Sidley 提供本信息仅作为向客户及其他友好人士提供的服务,且仅供教育目的使用。本信息不应被解释或依赖为法律意见,亦不构成律师与客户关系。读者在未寻求专业顾问意见之前,不应依据本信息采取任何行动。Sidley 和 Sidley Austin 指 Sidley Austin LLP 及其关联合伙实体,详见 www.sidley.com/disclaimer。
© Sidley Austin LLP
联系我们
如果您对本次 Sidley 更新有任何疑问,请联系您平时合作的 Sidley 律师,或
Offices
Capabilities
Suggested News & Insights
- Stay Up To DateSubscribe to Sidley Publications
- Follow Sidley on Social MediaSocial Media Directory

